Big Enough To Cover All Your Needs.
Small Enough To Care.
Discover how Biomapas provides all the support to accelerate your clinical trials and optimize your drug development process.
Clinical Research
Reliable support for every phase of your clinical trial, from recruitment to results.
Pharmacovigilance
Flexible safety monitoring to keep your products compliant and trusted.
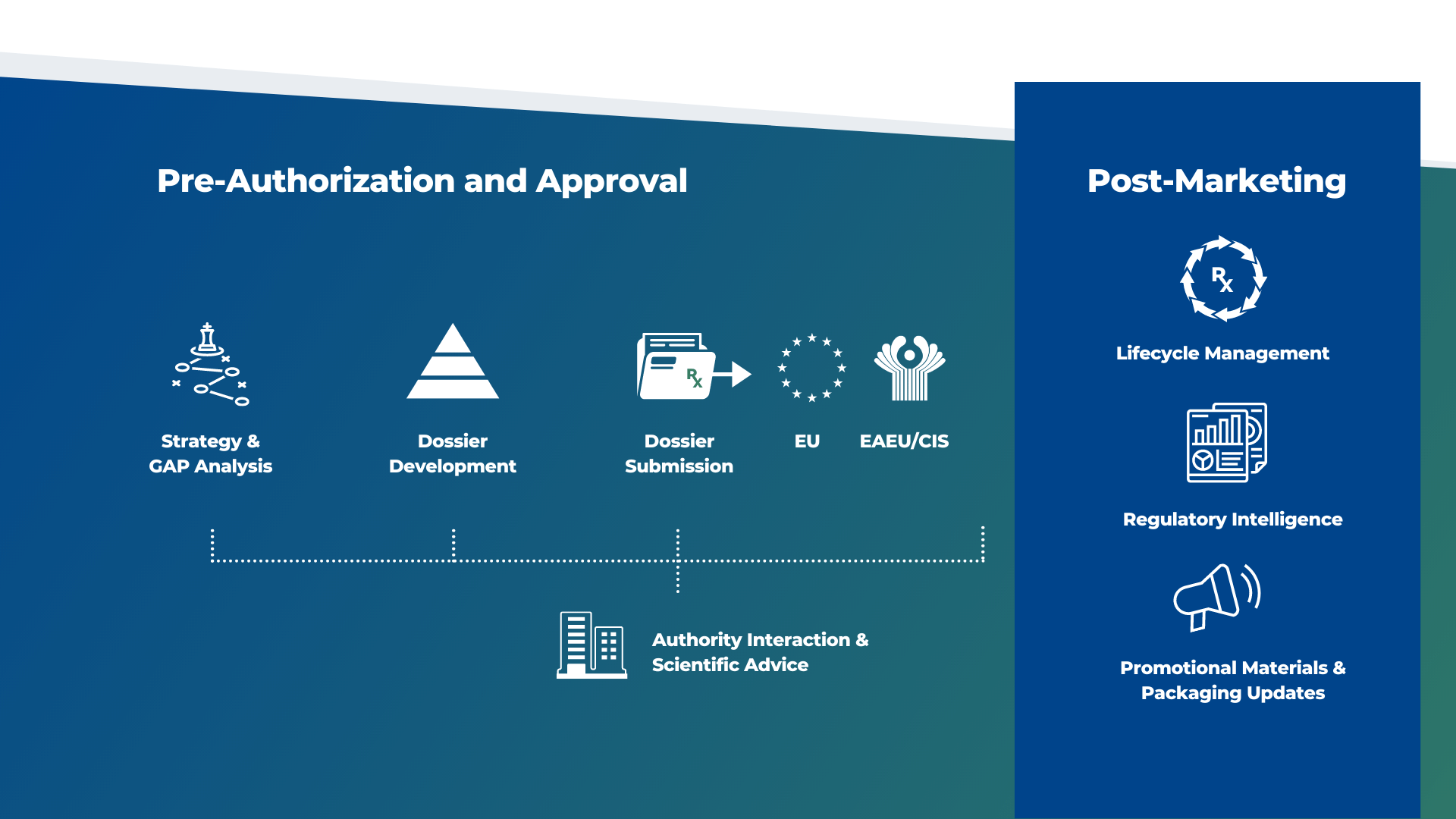
Regulatory Affairs
Confidently navigate regulatory challenges with expert support.
Medical Information
Clear, accurate medical information 24/7, tailored for global audiences.
Quality Assurance
Export auditors help identify gaps and close them with actionable solutions.