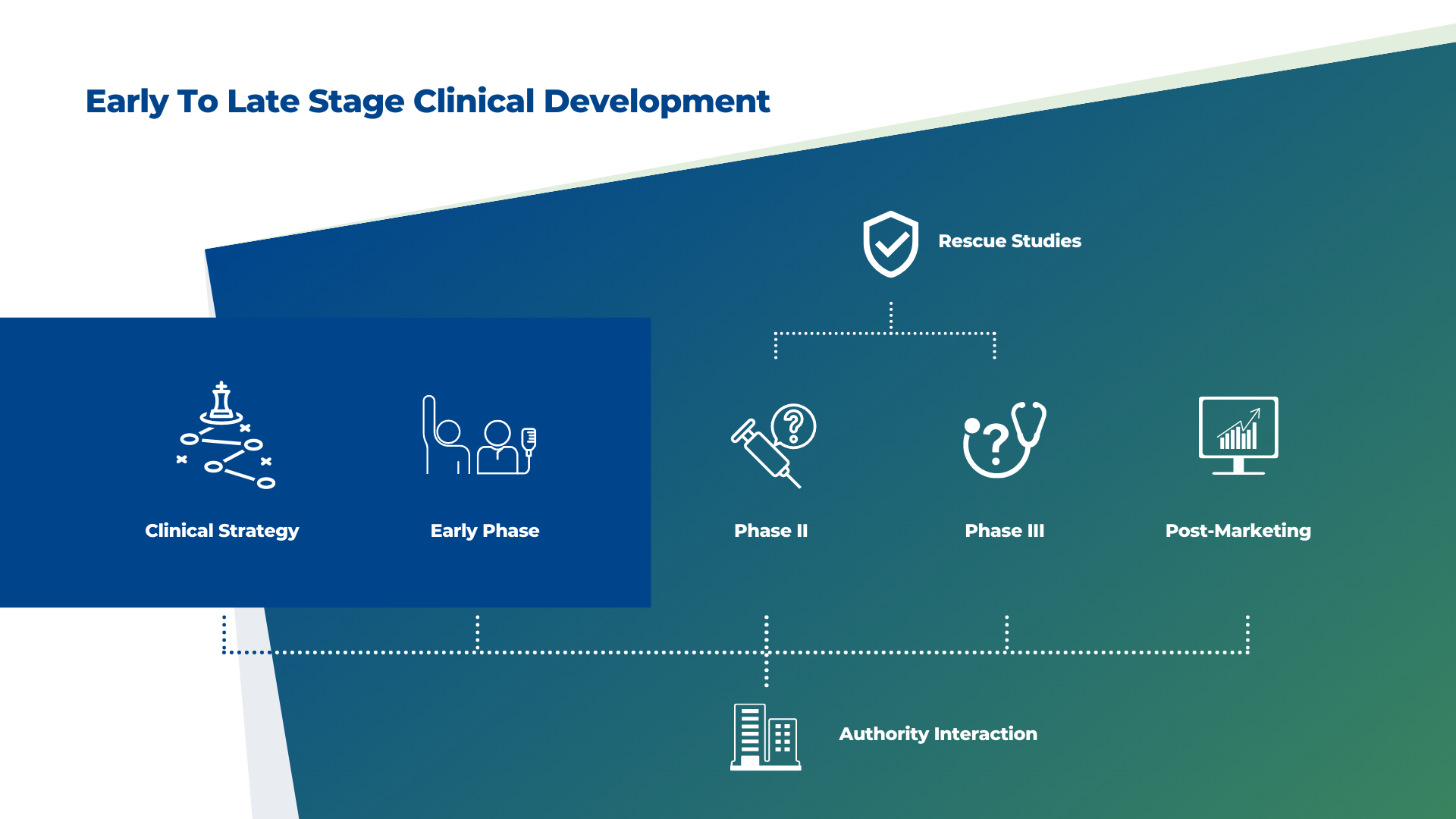
Clinical Strategy
For your trials to be successful, you need to be well-versed in science and technology. Knowing there is no one-size-fits-all approach to clinical development, we apply an integrated approach to development that leverages our different in-house disciplines. Biomapas' goal is to help you face increasing R&D costs, shorter product lifecycles, and embrace digital transformation in clinical development.
Early Phase
Rapidly progressing through early development to achieve proof of concept is crucial to your drug development strategy. It is also a vital milestone of hope for patients with unmet needs.
Biomapas’ experts help to shorten early clinical trials as a development stage while ensuring the integrality of all documents. Our early development teams combine forces with our regulatory and pharmacovigilance experts to enable informed decisions concerning, e.g., drug dosages, safety selection, PK/PD analysis, and design efficient study protocols.
We use our multidisciplinary expertise to generate all data required and turn it into meaningful strategic decision-making information.
Phase II
To uncover the full potential of your therapy, we craft precise recruitment strategies, engage expert medical writers, and streamline clinical site management, ensuring your study progresses efficiently toward proof of concept.
Phase III
As you prepare for market approval, our team ensures large-scale patient recruitment, robust data management, and seamless collaboration with key opinion leaders, guiding your therapy through the final and most critical phase.
Post-Marketing
Medicinal product lifecycle management is defined as the process of managing the entire lifecycle of the product. This includes its clinical development, marketing authorization and post-authorization activities. As medicinal products that are already marketed change over time, expertise in renewals, variations, and other medicinal product lifecycle management activities is one of the key elements to reach the company’s business goals.
Rescue Studies
Whatever the situation, Biomapas can help you finish your studies on schedule, improve the quality of your trials, or have your research approved again by a regulatory body.
Your research duration, regulatory filings and ultimate commercialization are all affected by these variables. As a result, after evaluating the extent and kind of study management deficit and identifying the sort of repair needed, we may act as a third-party function.
Authority Interaction
During drug development, marketing authorization, or later in the medication's lifecycle, interactions with health authorities may impact your product's outcome and financial success. Meetings with health authorities allow you to fine-tune your development plan with feedback from potential marketing authorization decision-makers. Biomapas prepares you to get the most out of them and informs you of the authorities' priorities, questions, and expectations.


Expertise in Managing Complexity
We adapt to the unique requirements of your trial, from complex early-phase oncology prototypes to high-volume, late-phase studies. Bioequivalence studies? We’ve got you covered, delivering cost-effective, high-quality results with proven expertise.

Access to Hard-to-Reach Patient Populations
Recruitment can stall a trial, especially when targeting underrepresented populations. We help you access diverse, trial-naive patients through underutilized sites across Europe, CEE, and CIS regions.
Flexible, Personalized Support
Every trial is different, and so are your needs. We adjust to your project, offering tailored support—from full-service management to specific tasks—working seamlessly as an extension of your team.

Related insights

Clinical Trial Diversity

Blog
Find out how, Functional Service Provider models became indispensable for the pharmaceutical and biotechnology sectors.

Free Guide
The guide is intended to serve as a useful resource for sponsors and investigators to navigate the various aspects of clinical trial conduct and to provide guidance on how to comply with the requirements of the CTR.

Insights delivered to your feed?
Sign up for the Biomapas Bulletin on LinkedIn






